


The annual reports are broken up into sections. Click on the tab heading to view the report section submitted by the respective section investigation leader.

California: Mike Devencenzi, Joseph Grant (UCCE), Jed Walton, Dr. Marshall Johnson (UC Riverside, outside scientist), Carolyn Pickel and Walt Bentley (UCIPM), Drs. Pete Goodell, Lucia Verela and Tunyalee Martin (UCIPM)
Oregon: Rich Garvin, Bruce Decker, Jeff Olsen (OSU Extension)
Washington: Dan Flick, Nick Stephens, Karen Lewis (WSU Extension), Dr. Doug Walsh (WSU, outside scientist)
Canada: Dr. Gary Judd (Agriculture and Agri-Food Canada, outside scientist)
Our project has just finished the fourth year of five. We have finished all the field work and nearly all the laboratory work and are in the process of analyzing, synthesizing, writing, and making outreach presentations.
One of our major accomplishments this past year was the two-day biological control (BC) short course that were held in three locations by teleconference on 7-8 February. We had 75 attendees, and 11 speakers from California, Washington, and Oregon covering 15 topics on basic and applied aspects of BC in orchards. The course consisted of presentations, case studies, and interaction with the instructors. It was followed up with a natural enemy identification and sampling course presented at field days at Hood River and the WSU-Sunrise orchard in Wenatchee in August. Information from the courses is on the web site, including narrated presentations from the short course. Attendees were enthusiastic the course (see Obj. 6).
Our web site (enhancedbiocontrol.org) is also undergoing continuous upgrades as the field and laboratory research is being completed, and we are adding video stories describing the methods and results of our research. The web site was re-done this year to allow content to be viewed on desktops, laptops, and tablet/smartphones (see Obj. 6).
A major goal for our project this coming year will be the publication of 14 scientific articles in the journal Biological Control as a special issue. These publications will cover all aspects of the project from beginning to end, and will include laboratory and field results as well as our successes and failures in outreach. Working with a high quality journal like Biological Control ensures that the information will reach a broad audience and that it is available both on-line and in larger libraries for the foreseeable future (see Output).
Finally, we are still aggressively pursuing funding that will allow us to proceed with logical extensions to our project. We have leveraged the funding provided by USDA-NIFA with another $1.2 M (approved or under review) from various sources since the start of the project (see Output).
Although the project directors are ultimately responsible for the work done in this project, there is also a key group of post-doctoral research scientists and technical support personnel that have been essential to our project through their dedication and hard work. We gratefully acknowledge their efforts to make the project a success:
Complete laboratory bioassays for all pesticides and 8 natural enemies and completed second year of field studies.
All bioassays have been completed except three sublethal assays for the ladybird beetle, Hippodamia convergens. Those assays were delayed until the reason for high immature mortality was tracked down and eliminated. We expect those to be completed in the next 3 months. All field trials were completed last year, and synthesis and presentation of the data are currently in progress.
We had significant problems with the survival of immature H. convergens that were unrelated to pesticide treatment. That problem has been solved and four of the seven sublethal assays have been completed. The current summary of lab studies is found below. Significant progress has been made in the analysis of the sublethal effects and all of the work that has been completed has been analyzed. We are also working with the web group (Objective 6) on how to present pesticide effects in a way that is intuitive and allow the interested person to “drill down” to specific causes of reduced population growth. This data will also be combined with a large pesticide effects database compiled by Dr. Ute Chambers (Objective 6) from literature sources and we will strive to provide the most up-to-date information on our web site.
The lab bioassays appear to show differences between predators and parasitoids (green section below). The combination of the phenology of natural enemies (Objective 2) and the natural enemy susceptibility will provide us with powerful tools to decrease adverse effects on biological control in western orchards. It is clear in some situations we will have to choose which natural enemy to preserve. Further synthesis this coming year will attempt to make a decision tree that can be used to guide management tactics.



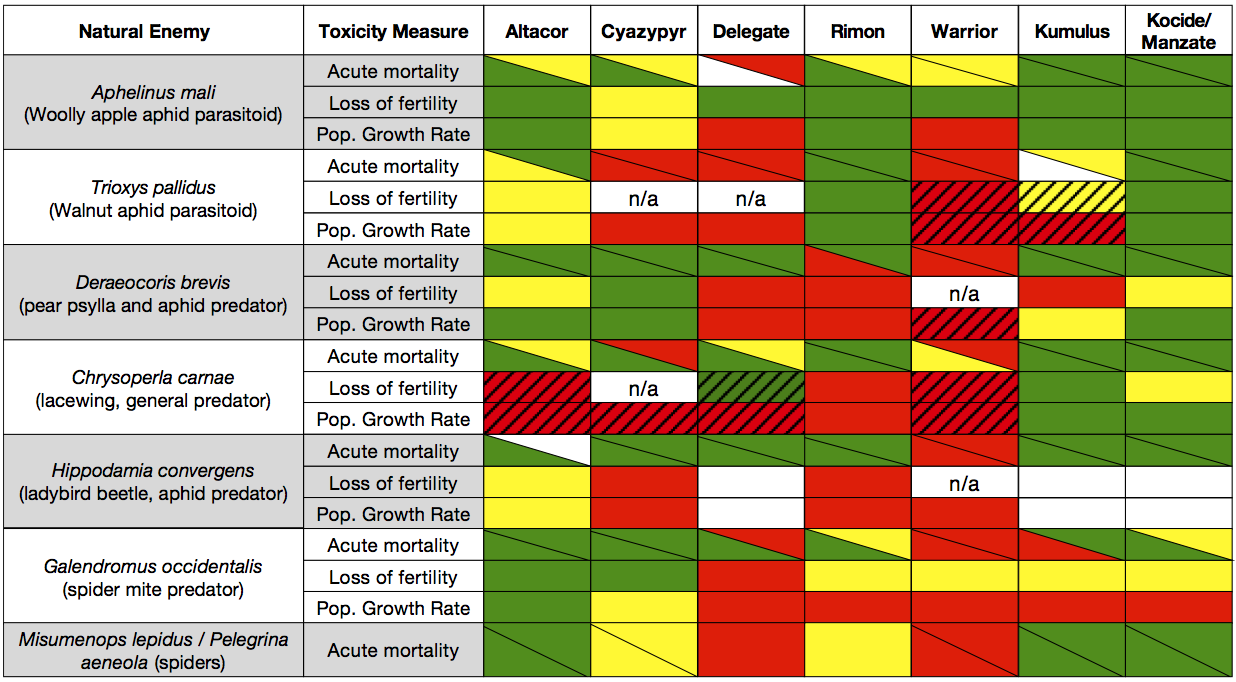
The chart above shows the current status of the lab bioassays. The population growth rates are probably the best indicator of pesticide effects until we finish some of the modeling efforts discussed in last year’s progress report. By assigning a value of 0 for green cells, 1 for yellow, and 2 for red, a very simple index allows us to broadly categorize the effect of each pesticide on each natural enemy. The most interesting comparison in the chart is to evaluate whether predators respond differently than parasitoids to different pesticides.
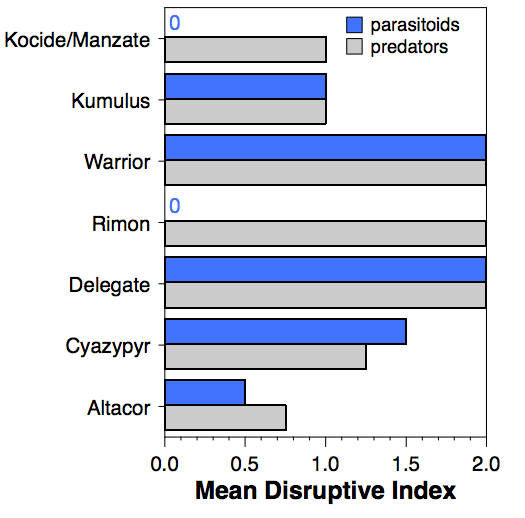
Delegate and Warrior both show the greatest potential for disruption of biological control (fig. 1). Cyazypyr and Kumulus both had moderate effects on predators and parasitoids, while Altacor had the lowest effect on both groups. Rimon had a large impact on predators while Kocide/Manzate had moderate effects on predators, but both insecticides had very low effects (<25% reductions) on the two parasitoids.
The differences between the predators and parasitoids are probably caused by the differences in their life history. Predators are free-living and are exposed to the pesticide residues for their entire life, whereas the two parasitoids we tested are endoparasitoids and thus the immature stage is completely protected within the aphid host.
Completed analysis and development of four different natural enemies that occur in apple, walnut, pear, and sweet cherry.
This section exceeded the milestones and goals of the grant. We are still collecting research data using our leveraged funds that will provide additional models and validation information.
The data in hand from apple, pear, walnut, and sweet cherry allowed us to develop models for two species of lacewings (Chrysopa nigricornis, Chrysoperla plorabunda), a syrphid fly (Eupeodes fumipennis), and the mirid bug, Deraeocoris brevis. (See Making a NE Model below.) All of these natural enemies are important generalist predators contributing to suppression of multiple species of aphids, pear psylla, spider mites, small lepidopterous larvae, mealybugs, and other soft-bodied insects and mites.
Our data set included not only the information on temperature and abundance of the different natural enemies, but also when pesticides were applied throughout the season. Our analysis showed that some of the pesticides strongly disrupted natural enemies and that normal early spray programs (especially lime sulfur + oil) in apple, pear, and sweet cherry are applied multiple times as three of the four natural enemies we studied emerged in the spring. These applications thus expose the adults throughout their emergence period, and the eggs and immature stages of the first generation. The standard supposition has been that these sprays occur when natural enemies are not present, but our research shows otherwise. (See NE Phenology by Crop below.)
We will be working with pathologists and horticulturists to see which sprays can be changed to non-toxic alternatives or moved to either late fall or earlier in the season before natural enemies emerge. These changes should boost natural enemy population levels and reduce secondary pest problems that have been an issue as our management programs have moved to newer insecticides.
We are using traps developed in objective 3 to monitor the natural enemies in a consistent manner throughout the entire season. Traps are checked 1-2 times a week and the number of each natural enemy species recorded. These raw data ares then combined with temperature records taken either in that specific orchard or within only a short distance. We also collect the spray records for each orchard so that any deviation in the phenology can be traced back to pesticides applied or application timing. With data from multiple locations in each state over a period of three or more years, we are able to generate a model in a short amount of time.
Development of a model requires data on how fast a natural enemy completes development. Insect developmental rates are directly related to the environmental temperatures they experience and can be modeled using heat units called degree-days (DD). Most of the time this information can be obtained from the literature or from work specifically done in the laboratory of one of our investigators. If the developmental data are comprehensive enough, we can determine the lower threshold for development and start to evaluate phenology in the field data. If the literature or laboratory data are very cursory, we will have to estimate the developmental thresholds using our field data.
To generate the model, we start by separating the data into two groups, one used for model generation and one used for model validation. We normally developed the models using data from the studies in apple, and then used the rest of the data to evaluate model accuracy. In situations where the data set was not large enough across multiple sites/crops, we either break it up by year or acquire supplemental data collected from some of our leveraged funding.
The models we developed all are based on fitting a Weibull distribution to the field data. This was done using a programmable statistical package that allows us to assess fit, and then graph the results with overlays of the predicted emergence period. Further graphics allow us to overlay spray program information to assess spray impacts on phenology.
All the models need to be tested with data independent of the information used to generate the models initially. Comparing the model fit with independent data allows us to evaluate the strengths and weaknesses of the models and the scope of their applicability.
In developing the models, one of the things that jumped out at us is how sprays at certain times affected model performance. Each year we evaluated phenology in a minimum of three orchards per crop and often considerably more. For each of these locations, spray records allow us to evaluate how natural enemies are affected by different spray timings and pesticide used. These data will be supplemental to the field tests done in Objective 1, and will encompasses a much broader range of pesticides and timings than the controlled experiments in that objective. This will require a larger analytical effort, and will likely give less precise conclusions, but the results will be based on work in commercial orchards and better reflect spray programs normally used. This is an area we will be pursuing in the coming year.



Our data were collected across a wide geographical area so that climate, regional, host availability, and spray program differences are reflected.
Across four insect models, it is apparent that not every generation possible occurs at all locations. For example, we found that C. carnea only went through two generations per year in California, despite having enough heat to complete four generations. Washington had the greatest number of generations of C. carnea, with three full generations being completed. The syrphid fly, E. fumipennis was even more variable, with some locations recording only a single generation and others up to four (fig. 1); differences between sites may also be related to the spray programs or host availability.
The Hood River locations recorded the fewest number of generations of all species, because seasonal temperature profiles there were considerably cooler than our sites in California or Washington.
Spray programs vary dramatically between crops, with few sprays being applied in walnuts, and a large number in pears. Apple and sweet cherry programs were highly variable, with greater differences between orchards than between crops.
All three fruit crops have very intense programs early in the season and tend to be very moderate later in the season. Different spray programs can result in the apparent loss of a particular generation for some natural enemies.
Natural enemy species abundance varied dramatically between the three regions. Some of the differences were likely caused by simple temperature effects, but also by surrounding vegetation and crop systems. Washington orchards had dramatically higher lacewing populations than the California or Oregon orchards surveyed. The apple, sweet cherry, and walnut orchards all had relatively abundant and diverse syrphid fly faunas, while the fuana in pear was considerably less abundant because aphids tend to be rare in pears.
Variations in phenology between crops were no greater than variability seen between locations within a crop. Although climatic differences in California may be causing diapause or reduced populatio levels during the heat of the summer with C. carnea, we do not have evidence of crop effects on phenology (versus climatic, pesticide effects or host abundances).
We have completed studies on lure longevity, optimal release rates, trap types, and mixtures versus separate lures, with >65 attractant blends.
This section exceeded the milestones and goals of the grant. Summary and synthesis of this information will continue for the next 1-2 years.
All field studies have been completed, so we are in the process of a complete analysis of the different regional studies. One of the issues we face is that often we get no trap catch in our unbaited control traps, which confounds the statistical analysis. This also occurs with acetic acid traps for some of our natural enemy groups, but when acetic acid is added to another attractant, we get synergistic effects. Thus, a great deal of time is being spent trying to choose the best statistical analysis, even though the results are generally straightforward. As discussed in the progress report last year, we can tailor our attractive blends to target certain natural enemies that are important in each of the systems.
We also worked with the Objective 6 group to put out grower/consultant trials in Washington and Oregon of the squalene lure, which attracts the lacewing Chrysopa nigricornis. These trials were to familiarize participants with the lures and compare different management tactics on the lacewing.
Our work is providing effective and simple sampling tools for natural enemies (figure below). We need to have more industry involvement with the development of commercially available lures. We hope to make more progress on that goal this year, but it is possible that this will require more time than is left in the grant.

Developed a rapid and robust method to evaluate content of arthropod predators. Evaluated over 2400 samples to estimate predation rates of likely codling moth predators.
Samples analyzed this past year have clarified the ecological role of three different predator groups. The work on DNA preservatives used in pitfall traps has resulted in better specimens being collected and new PCR primers in 2010 allow the amplification of codling moth DNA without requiring purification. These results are a major advance in our ability to evaluate food chain relationships between different predators and prey species.
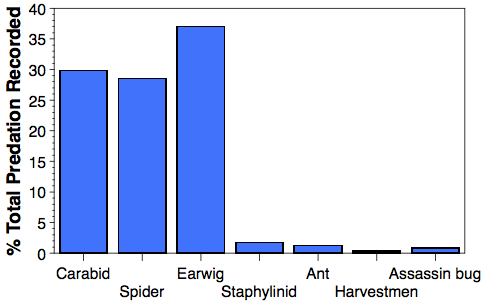
Our samples collected from seven apple orchards over a three year period showed predation rate on codling moth was 9.2%. We also found that a remarkable 95.5% of the total predation could be accounted for by just three predator groups: ground beetles (Carabidae), the European earwig (Forficula auricularia) and the spider complex (Araneae) (graph 1). Predation rates across orchards generally correspond to management intensity; in orchards with lower management intensity, abundance of ground dwelling predators, particularly carabids, is high.

Predation of codling moth on the ground appears to be dominated by earwigs, predacious ground beetles, and spiders. With the newer high density apple orchards, more codling moth are likely to overwinter in the ground cover (versus on the tree) because there are fewer overwintering sites on the smooth bark in the younger orchards, thus our knowledge of ground fauna and their impact on codling moth will help shape future management programs.
The whole idea behind DNA gut content analysis is that we can collect various predators and evaluate if they have fed on codling moth by using PCR to look for specific sequences of DNA found only in codling moth. While this is straightforward, the problem is that the DNA is only detectable for a relatively narrow window in time after the predation event. The length of time is a function of the length of the DNA sequence the primers are designed to detect. The longer the DNA sequence, the quicker it might be destroyed by the digestive enzyme in the predator. In addition, because insects’ metabolic rates are related to the temperature that they experience, the rate of
degradation is quicker in the warmer times of the year, which reduces the time over which the target DNA sequence can be detected. There are also differences in the digestive rates between different groups of natural enemies (e.g., spiders versus ground beetles), which also confuses the interpretation of the data. Thus, while we might only detect 10% predation from a particular group, that number only provides a snapshot of what happened within a fairly narrow window (<3 days) in time. To provide an estimate of the predator assemblage impact on codling moth, we need to take long time series of samples and integrate the effect over each generation of codling moth. This analysis is ongoing.
Synthesize information on growers’ willingness to pay for indirect benefits of IPM, develop an expected profit model using enhanced BC and synthesis for use in Objective 6.
This objective is on track to meet the goals and milestones of the grant.
Pesticide cost analysis showed pear IPM costs Oregon pear growers an average of $811/acre, compared to $288/acre for Washington apple growers. Most of this difference in cost is for control of pear psylla (see below). Unlike the apple analysis, we found no relationship between codling moth control programs and disruption of pear psylla biological control. Instead, pear psylla programs appear to be the biggest disruptor of biological control of psylla and spider mites.
Analysis of walnut data likewise showed no relationship between use of highly disruptive insecticides and costs of control for secondary pests.
Analysis of the apple data last year suggested that there was a straightforward relationship between the the use of pesticides disruptive to biological control and the cost of management for secondary pests such as aphids and mites. However, when running the same analyses on pear and walnut, there were no differences detected between orchards that used highly disruptive or non-disruptive insecticide treatments. What makes the pear and walnut systems different and can we capture the value of biological control in those systems?

The pear system is distinctly different from apple and walnut because pear psylla is the driving force behind much of the management program and psylla sprays are often applied prophylactically. Psylla is considered to be an induced pest that would normally be controlled by predators, if the predators were not suppressed by insecticides applied for psylla or codling moth.
A second major difference is that biological control of mites is much more tenuous in pear because even low level mite feeding can result in leaf necrosis. This sensitivity to mite feeding leads many growers to apply miticides in a tank mix with insecticides targeted at pear psylla. The third major difference from apples is that aphids are relatively rare in pears, probably because the insecticides used in control of pear psylla tend to be very effective against aphids as well.
The walnut system is also considerably different than the apple system. In walnuts, many of the insecticide treatments occur later in the season and are targeted against later generations of codling moth and against the walnut husk fly. Walnut aphid is both an early and late season pest, with reduced growth during the summer. Thus sprays in the early season might be disruptive, but those in the summer are much less likely to cause aphid outbreaks. Early season control of codling moth is also not as much of an issue in walnuts, so again, early season treatments are not as common, which gives the parasitoid Trioxys pallidus and generalist predators a better chance to suppress walnut aphid numbers. Spider mites can be an issue in walnuts, but again tend to be a later season pest, so disruption early in the season would play a large part in outbreak scenarios. Finally, the large size of walnut trees makes it much harder to get uniform coverage of pesticides, particularly in the upper canopy. This results in the upper canopy acting as a partial refuge for natural enemies and likely gives a more stable predator/prey interaction than would be found in systems where spray coverage is more uniform.
Use survey results to guide development of educational and outreach programs, synthesize data from completed objectives and implement into management programs, present results to industry.
This section has met and exceeded the goals of the grant in many aspects.
Our group has begun the various outreach programs as information has become available. We have offered short courses, workshops, given presentations at numerous grower meetings and field days. Our team has updated and developed new handouts on our progress, identification of key natural enemy groups and synthesized literature information on pesticide impacts on natural enemies (see below). Team members have collaborated with industry publications (Good Fruit Grower, Capital Press, and Western Farm Grower) to provide information on various parts of the project. We have also conducted real-time surveys at grower meetings to evaluate where growers and consultants obtain information for pest management and their attitudes towards biological control and barriers to its' implementation. (below)
Using an audience response system (“clickers”), we conducted surveys of grower/managers and crop consultants working with apples at four winter meetings in north-central Washington. The surveys captured how industry stakeholders obtained information about pest management and biological control, and their use of conservation biological control. We surveyed 207 were grower/managers and 77 crop consultants. summaries of these will be on our web site shortly.
The top three ways Grower/Managers wanted information were in person meetings/courses (57%), field days (48%), and printed materials (42%). Consultants wanted information by in person meetings/courses (60%), website resources (51%) or email (47%). To optimize information flow, we must address both groups' preferences.
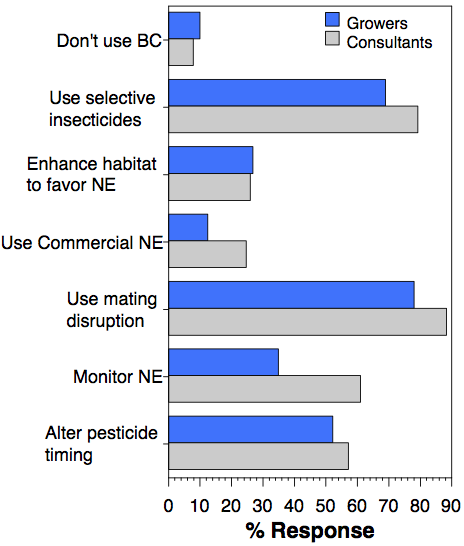
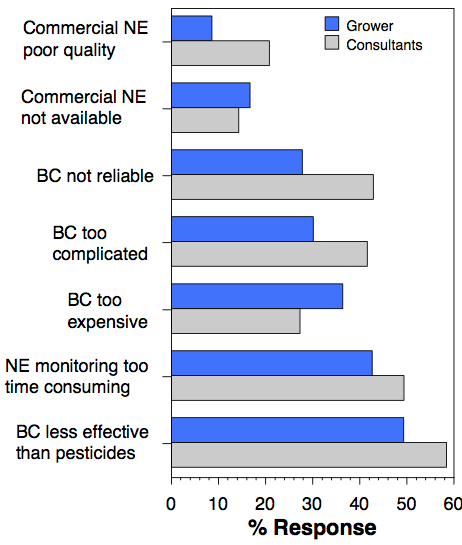
Successful integration of biological and chemical control requires a better understanding of the crop systems and how different tactics affect natural enemies.
In February 2012, our project members, advisory group, and colleagues presented a comprehensive course highlighting the information needed to use and implement biological control in orchard systems. The course was offered simultaneously to 75 participants in Washington in Wenatchee and Pasco, and in Oregon at Hood River via teleconferencing. The majority of the presentations from the course as well as participant feedback are available on our website at enhancedbiocontrol.org (see BC Short Course under "Features" on our site menu).
Day 1
Day 2
A single presentation isn’t enough to effect change in the industries. In addition to the two-day short course, we supplemented our course with lab and field presentations featured at the OSU Hood River Experiment Station field day on 3 Aug and the WSU Sunrise Field Day on 23 Aug. Participants received high quality color handouts of the major natural enemy groups and identification tips using microscopes (handouts are found online here). Once they finished the lab work, we moved to the field, demonstrated various collection techniques and then had the students collect and identified the different natural enemy groups. More of these short identification workshops will be held this coming season to broaden our audience and help develop a core of people with understanding of the various natural enemies and the pests they control.






As our project enters the final year, we are concentrating resources to leave a legacy of web resources describing the findings of our project. Normally, researchers have an incredibly annoying habit of moving on to new projects as they finish old ones, but in our case, we know that we need to preserve, organize, and provide the information generated in the project. We have already provided narrated slide shows from our short course, and are working to get other presentations given by project members up on the web. Three YouTube videos of methods used in the project are already on-line and at least four more are in production. We are also making a complete archive of stories written by various media sources about the project, publications associated with the project, and both photo and video galleries of natural enemies in action. The new web site is also usable with tablet and smartphones as well as the normal desktop/laptop computers. enhancedbiocontrol.org
We have performed done a survey of pesticide effects on natural enemies in orchard systems. This has been compiled into a database, and will be available on-line sometime in the spring. This database synthesizes research on a broad range of pesticides (not just those evaluated in this project) done in Europe, California, Oregon, Michigan, New York, Ohio, Pennsylvania, and Washington giving users a broader perspective about natural enemy effects across a wide range of conditions and locations.
WSU-DAS allows us to share in real-time information on IPM tactics, models of pests and natural enemies, pesticide choices that minimize natural enemy mortality, and provide timely stories important for IPM decision makers. DAS will feature prominently in the outreach within Washington state as we finish and validate the models for the lacewings Chrysopa nigricornis and Chrysoperla carnea, the syrphid fly, Eupeodes fumipennis, and the predaceous bug, Deraeocoris brevis (Objective 2). These models and the spray recommendations (fig. 1) will be crucial in enhancing biological control in our apple, pear, and sweet cherry orchards. We will also provide the information to our colleagues in California and Oregon, as well as provide the information on our project web site. The new version of DAS is fully HTML 5 compliant and will allow iOS and android smartphones to access to all features of the web site. das.wsu.edu
While web and popular articles are important, our project members are committed to publication of our findings in high quality peer-reviewed journals. Towards this end, we have contacted the journal Biological Control and arranged for the publication of a special issue with 14 different articles (titles listed below) all by our team members. These articles will go through the normal peer review process and be published together in a single issue devoted to our project. This method of publication brings our work together in a single location available in the best libraries in the world as well as available on the internet. We are proud to work with Biological Control in this project and especially thank their editors and Dr. Ed Lewis (UC Davis) who has agreed to serve as the coordinating editor.
From Planning to Execution to the Future: An Overview of a Concerted Effort to Enhance Biological Control in Western Apple, Pear, and Walnut Orchards
Jones VP, NJ Mills, JF Brunner, DR Horton, EH Beers, TR Unruh, PW Shearer, J Goldberger, K Gallardo, S Castagnoli, N Lehrer, SA Steffan, KG Amarasekare, U Chambers, AN Gadino
Testing the Selectivity of Pesticide Effects on Natural Enemies in Laboratory Bioassays
Amarasekare KG, PW Shearer, NJ Mills
Comparative Analysis of Pesticide Effects on Natural Enemies in Western Orchards: a Synthesis of Laboratory Bioassay Data
NJ Mills, EH Beers, PW Shearer, TR Unruh, KG Amarasekare
Large-Plot Field Studies to Assess Impacts of Newer Insecticides on Non-Target Arthropods in Oregon Pear Orchards
PW Shearer, KG Amarasekare, EH Beers, NJ Mills, VP Jones
Nontarget Effects of Orchard Pesticides on Natural Enemies: Lessons From the Field and Laboratory
Beers EH, NJ Mills, PW Shearer, DR Horton, E Miliczky, KG Amarasekare
Evaluating Release Rates and Longevity of Natural Enemy Attractant Lures
Jones VP, CC Baker, AJ Bixby-Brosi
Optimizing Herbivore-Induced Plant Volatiles and Floral Volatiles for Monitoring of Key Natural Enemies in Apple, Sweet Cherry, Pear and Walnut
Jones VP, CC Baker, TD Melton, SA Steffan, NJ Mills, PW Shearer, KG Amarasekare, TR Unruh, DR Horton, E Miliczky
Using Herbivore-Induced Plant Volatiles and Floral Volatiles to Attract Natural Enemies for Studies of Ecosystem Structure and Function
Jones VP, CC Baker, SA Steffan, TD Melton, NJ Mills, TR Unruh, DR Horton, PW Shearer, KG Amarasekare A Bixby-Brosi, E Miliczky
Using Natural Enemy Lures to Develop Phenology Models for IPM Purposes: Examples from Lacewings and Syrphids
Jones VP, NJ Mills, PW Shearer, TR Unruh, DR Horton, E Miliczky, TD Melton, CC Baker
The Spider Fauna Using Codling Moth, Cydia pomonella, in Apple Orchards as Determined by Molecular Gut Content Analysis
Unruh TR, E Miliczky, DR Horton
Capturing the Economic Value of Biological Control
Gallardo K, JF Brunner, S Castagnoli, NJ Mills, J Grant
Biological Control Adoption in Western Orchard Systems: Results from Grower Surveys
Goldberger J, N Lehrer, JF Brunner, NJ Mills
Web-based Outreach for Orchard Management Decision-Makers
Jones WE, U Chambers, AN Gadino, JF Brunner, VP Jones
A Perspective on the Extension of Research-Based Information to Orchard Management Decision-Makers: Successes and Failures and potential future directions.
Gadino AN, JF Brunner, U Chambers, S Castagnoli, WE Jones